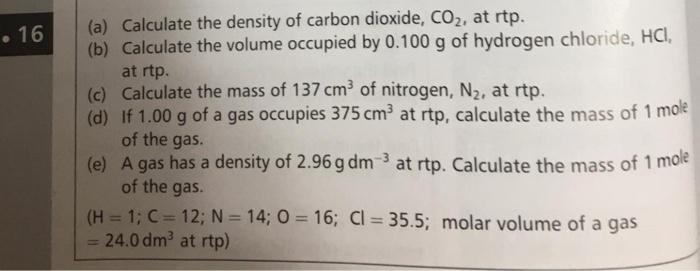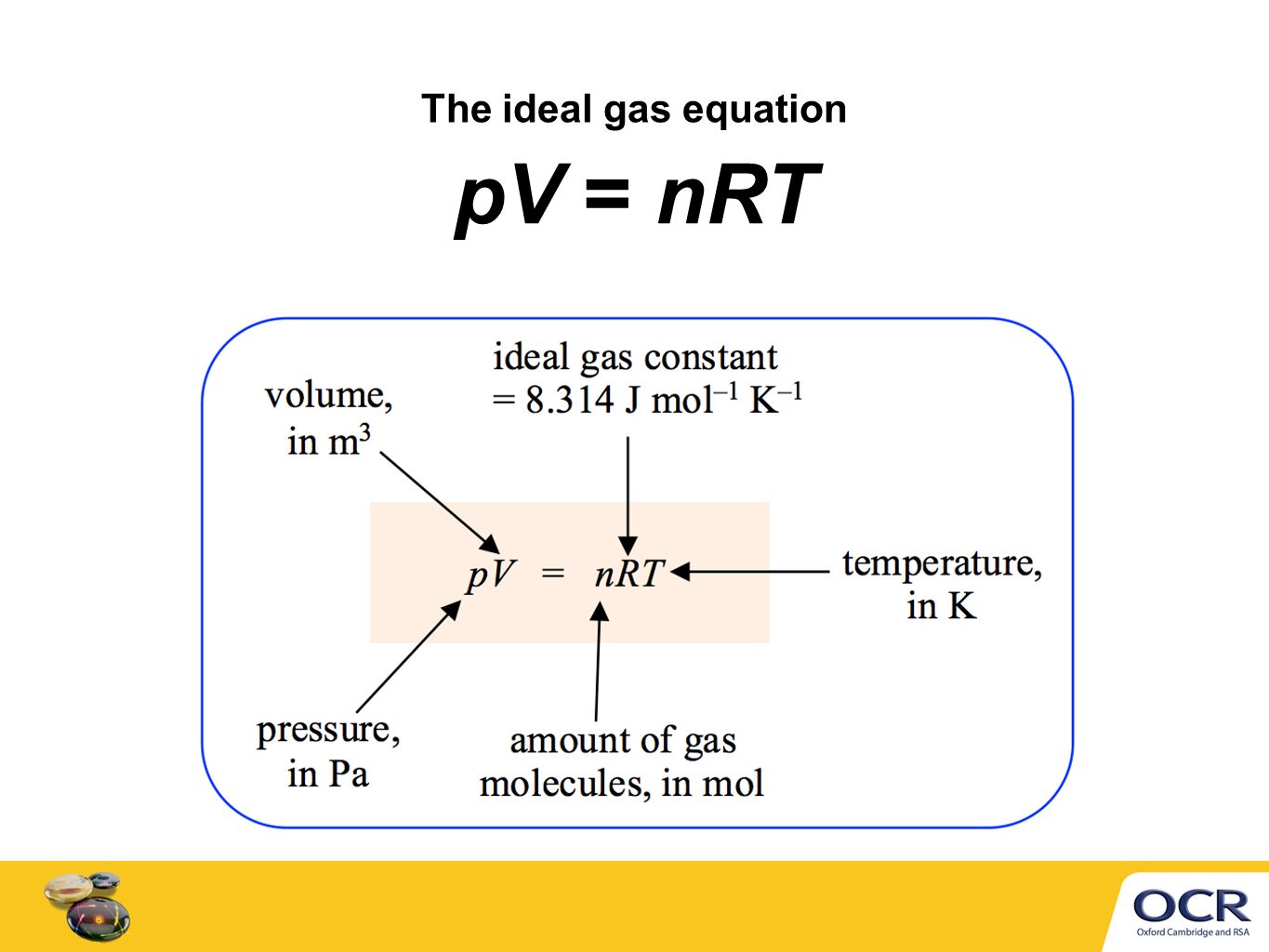
The ideal gas equation. Room temperature and pressure, RTP Limitations At RTP, 1 mol of gas molecules occupies 24.0 dm 3 Conditions are not always room. - ppt download
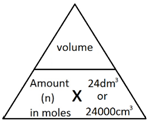
1:35 (Triple only) understand how to carry out calculations involving gas volumes and the molar volume of a gas (24dm³ and 24,000cm³ at room temperature and pressure (rtp)) - TutorMyself Chemistry
SAMPLE CALCULATIONS for STP and RTP ANSWER KEY Watch your sig figs! 1. How many moles of ozone occupy a volume of 3.36 L at STP

The ideal gas equation. Room temperature and pressure, RTP Limitations At RTP, 1 mol of gas molecules occupies 24.0 dm 3 Conditions are not always room. - ppt download

The ideal gas equation. Room temperature and pressure, RTP Limitations At RTP, 1 mol of gas molecules occupies 24.0 dm 3 Conditions are not always room. - ppt download
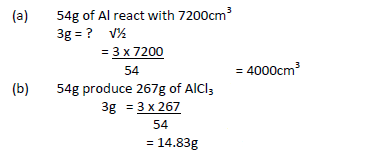
Given the equation for reaction below Calculate (i) Volume of chlorine at ( r.t.p) required to react with 3g of Aluminium (Molar gas volume at r.t.p =...

The ideal gas equation. Room temperature and pressure, RTP Limitations At RTP, 1 mol of gas molecules occupies 24.0 dm 3 Conditions are not always room. - ppt download

Calculate the volume of carbon(IV)oxide at r.t.p produced when 5.0 g of calcium carbonate is strongly heated.(Ca=40.0, C= 12.0,O = 16.0,1 mole of gas =22.4...